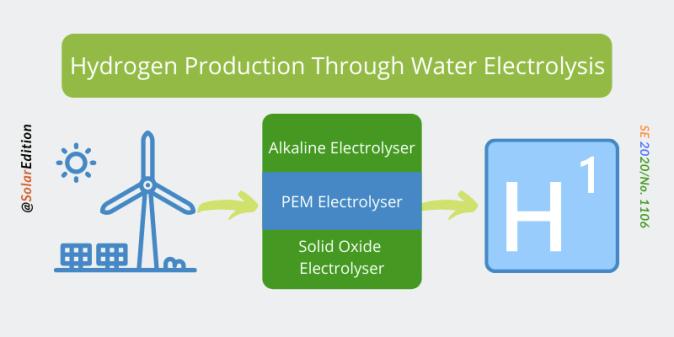
- Methods of making hydrogen The main processes for making hydrogen use natural gas, coal, or electricity with water
- Other options for hydrogen production Biomass, solar power and nuclear power could all be used to make hydrogen in the future
- The different ‘colours’ of hydrogen The environmental impact of making hydrogen depends on how it is made, and what it is made from
Although it is a very common element, there are no large reserves of hydrogen gas on Earth. This is because it bonds with other elements, so the hydrogen we need is locked up in other substances. By using energy (heat, electricity etc) it is possible to separate out the hydrogen from feedstockssuch as water and methane.
Today, over 95% of dedicated hydrogen production is from natural gas or coal, which produces greenhouse gas emissions. If we want to use hydrogen to help reduce greenhouse gas emissions, switching to a low-carbon method of production is required. The environmental impact of making hydrogen depends on what it is made out of (the feedstock) and the source of energy used to drive the process.
Current methods of making hydrogen
Hydrogen from fossil fuels
The most common method of making hydrogen today is called steam methane reformation (SMR), which combines methane (from natural gas) and water at very high temperatures (approximately 900°C) to produce a mix of carbon monoxide, carbon dioxide (CO2) and hydrogen. By controlling the amount of air, water and methane in the reaction, engineers can alter the SMR process and change the amount of energy required and waste gases produced.
Hydrogen can also be made from coal in a process called gasification. A syngas is created using coal and water at high temperatures. Above 750°C, the carbon in coal reacts with water to form a mix of gases, including hydrogen and carbon dioxide. The coal is used to provide the heat that the reaction needs to work.
Using fossil fuels to make hydrogen produces waste greenhouse gases. To make the process environmentally friendly, waste gases must be prevented from reaching the atmosphere by using carbon capture usage and storage (CCUS) technology, which can remove and store up to 97% of the CO2 emissions that are produced. However, CCUS is not widely commercially available at present.