- Why does hydrogen matter? We need to meet our energy needs in a clean, sustainable way. Hydrogen is a potential option, as it can be produced and used without releasing harmful emissions
- What is hydrogen? Hydrogen is a chemical element that can be burned or used in chemical reactions to provide energy
- What are the challenges? Producing, storing and moving hydrogen all require energy and resources and there are costs and risks involved
We use fossil fuels – coal, oil and natural gas – to meet most of our energy needs; to heat our homes, cook our food and power our cars. This has harmful impacts on our health, nature and the environment. Sustainable alternatives are required to meet our future energy needs and hydrogen could play a role in that new energy system.
Hydrogen is an energy carrier – it contains energy. Hydrogen can be used to produce energy with zero smoke, pollution or climate warming emissions – the only product is water. Producing hydrogen requires energy, but as long as it is made in a sustainable, low-carbon way, it is a potential alternative to burning fossil fuels. This could reduce the negative effects of greenhouse gas emissions and air pollution.
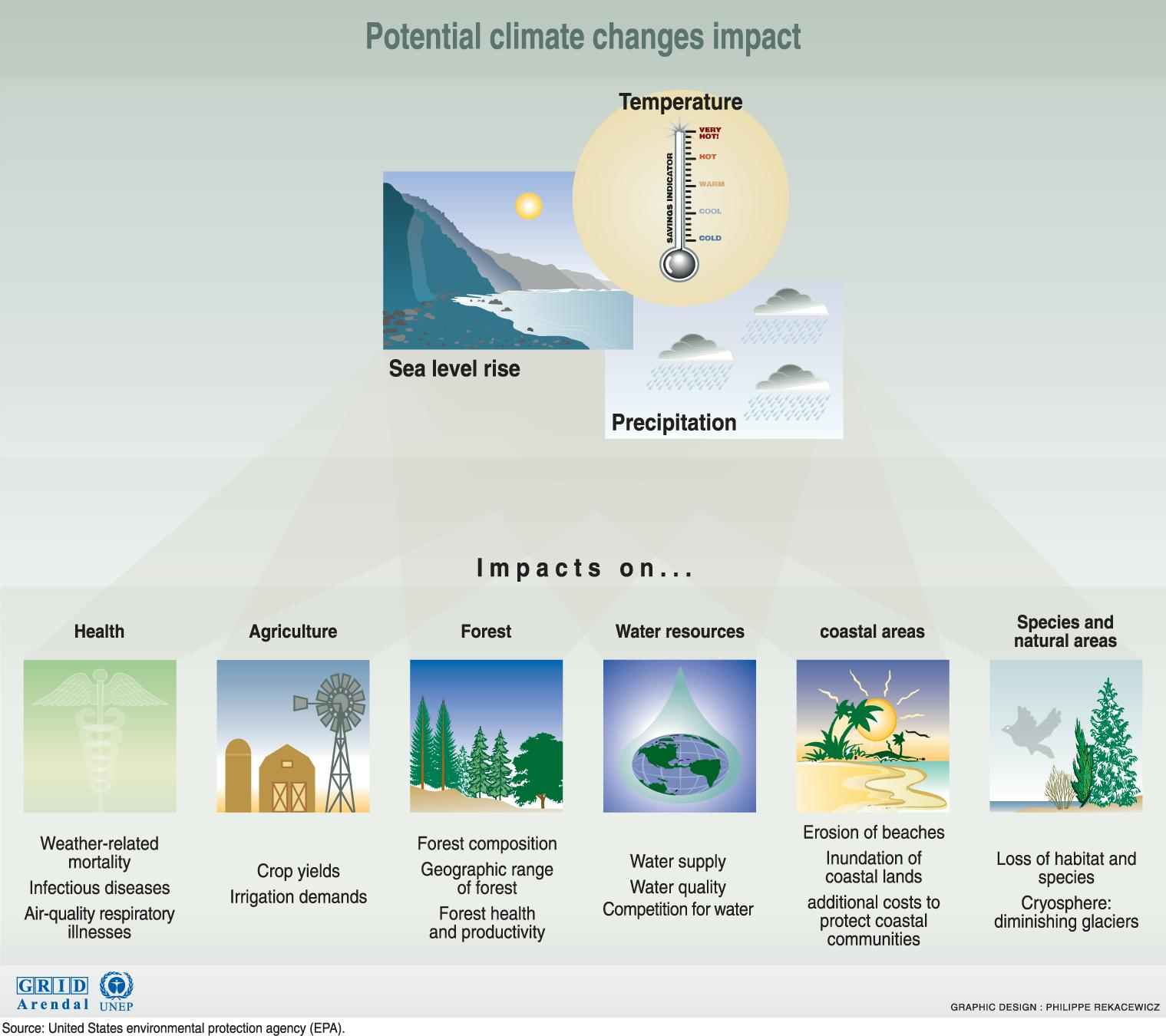
Extracting, processing and burning fossil fuels produce waste gases, including carbon dioxide (CO2), nitrogen oxides (NOx) and methane. As heat from the sun reaches the Earth, these gases trap extra heat in the atmosphere, which warms the planet. The glass roof of a greenhouse traps heat in a similar way, and therefore the gases are known as greenhouse gases. There is now overwhelming evidence that human-induced greenhouse gases are altering the Earth’s climate. This is known as climate change.
Burning fossil fuels also produces air pollution which can cause asthma, bronchitis and other health conditions. This is a particular problem in big cities, where exhaust fumes from homes, vehicles and power plants have a clear impact on public health.
What is hydrogen?
Hydrogen is a chemical element – one of the building blocks that all matter in the universe is made of. Of all the elements, it is the simplest and most abundant. Down on Earth, finding hydrogen gas in nature is very rare. However, hydrogen is reactive and forms chemical bonds with many other elements. As a result, there are huge amounts of hydrogen bound up in common substances such as water, methane and propane.
At normal room temperature and pressure hydrogen is a nontoxic gas; it has no taste, colour or smell, which makes it difficult to detect. If cooled to extremely cold temperatures (-253°C or lower) it becomes a liquid. Hydrogen is flammable and ignites easily in air, and it burns at over 2000°C with a very pale blue, near-colourless flame. When it burns, it reacts with oxygen in the air to produce water – the word hydrogen roughly translates to ‘water-maker’. Like natural gas, hydrogen can produce some NOx emissions when burned – however this can be managed with modern burner technology.

What are the challenges?
Just like any other energy carrier, there are challenges to using hydrogen; the main challenges are safety, cost, and making sure production is sustainable and low carbon. Hydrogen has been used in industry for many years but the vast majority is not produced in a low-carbon way – instead, natural gas and coal are used, which contributes to climate change. Storing and moving hydrogen around also require resources and involve costs and risks. Just like natural gas, hydrogen is flammable, so it must be treated with care to keep people safe.
If hydrogen is used in new ways to support a clean energy system, large volumes would need to be produced sustainably. Technology for producing low-carbon hydrogen already exists, but, at present, it is more expensive than other production methods or alternative fuels. Investment in new production facilities and supply chains would be required to meet higher demand and reduce unit costs.